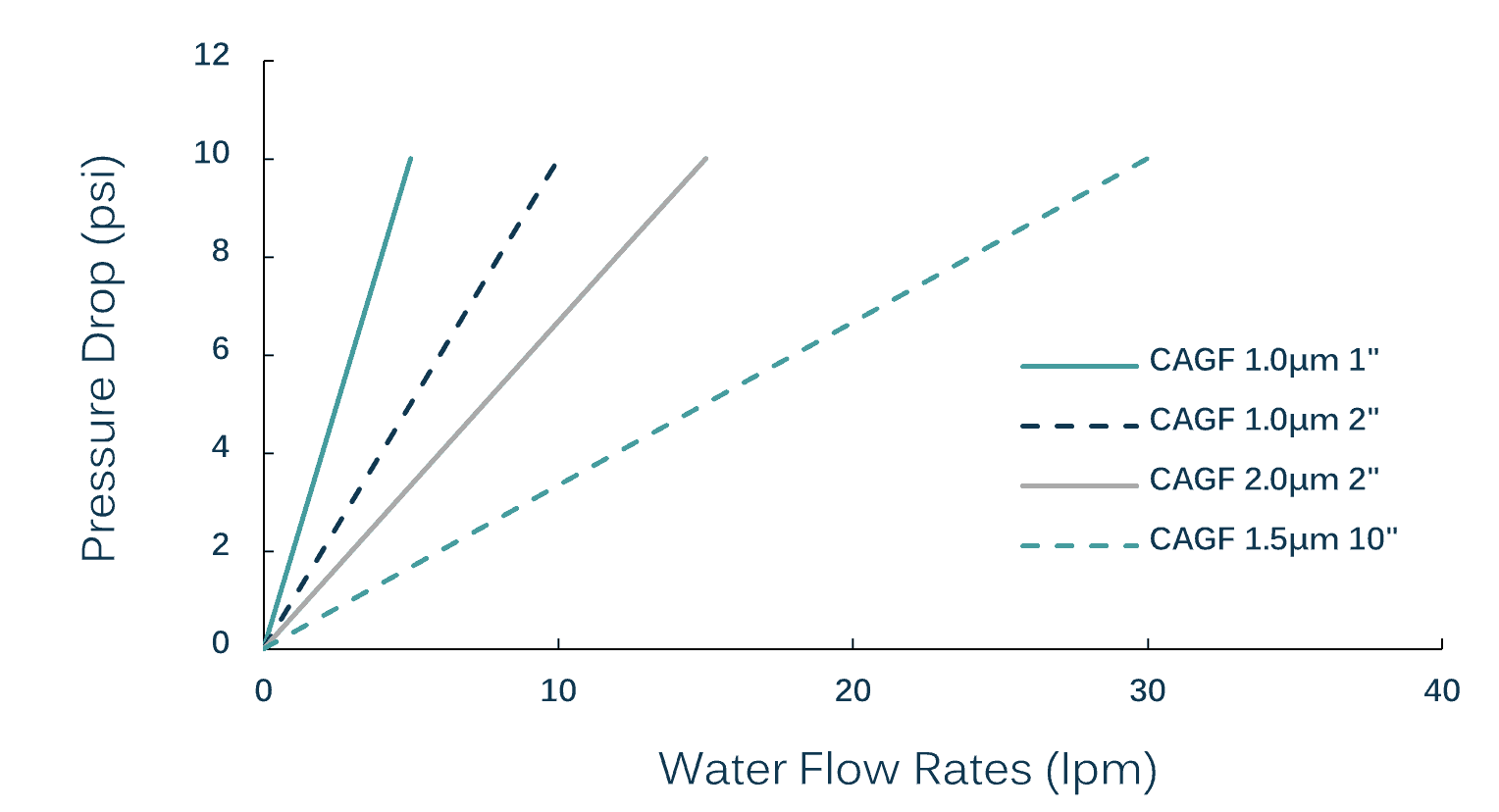Glassfiber Capsule Filters
The PoreCap® series of Glassfiber capsule filters are ideal for prefiltration and protection of downstream sterilizing grade filters and are especially designed for high particle retention, dirt holding and pre-filtration applications.
These are available in multiple pore size options ranging from 0.5µm to 2µm with 1-10 inch sizes. Each Glassfiber capsule filter is validated for flow rate, heat stability and integrity.
Membrane Characteristics: Our Glassfiber Capsule filters incorporate a polypropylene prefiltration layer and an upstream high dirt capacity glass fibre media layer suitable for removal of unwanted particulates.
APPLICATIONS
- Biological prefiltration applications
- Protection of final membrane filters
- Particulate/ coarse removal
- Pre-filtration of SVP & LVP
- Precipitate removal
- Water & aqueous filtration
Microbial Validation
Validated as per the ASTM F 838-05 requirements for determining bacterial retention of membrane filters used for liquid filtration
FDA Compliant
Biosafety & Extractables
Integrity Tested
Global Manufacturing Standards
Easy Identification & Tracking
Each filter unit has a unique identification marking which enables quick backtracking
100% Integrity Tested
Guaranteed Specification as per Actual Data
Non Leachable
Ergonomically Designed & Developed
TECHNICAL SPECIFICATIONS
Glassfiber Capsule Filters
| Materials of Construction | ||
| Membrane: | Microglassfiber | |
| Housing: | Polypropylene | |
| Core: | Polypropylene | |
| End Connections: | Polypropylene | |
| Rings: | Silicone (Only with 1/4" MNPT) | |
| Operational Parameters | ||
| Max Temp | 80 °C @ ≤ 2 Kg/cm2 | |
| Max Pressure | 4136 mbar (60psi) 30°C | |
| Autoclavable | 30 autoclave cycles of 30 minutes at 121 °C |
| Effective Filtration Area: | |||||
| Length: | 1″ | 2″ | 5″ | 8″ | 10″ |
| Area: | 0.025m2 | 0.05m2 | 0.10m2 | 0.20m2 | 0.60m2 |
QUALITY ASSURANCE & VALIDATION
All filters are 100% integrity tested and are developed and manufactured under an ISO 9001:2015 certified Quality Management System and designed to be used in cGMP compliant processes.
Microbial Validation
All our Cartridge and Capsule filters are validated as per the ASTM F 838 requirements for determining bacterial retention of sterilizing grade filters
Certificate of Quality
All our products are delivered along with a Certificate of Quality (CoQ) mentioning the materials of construction and testing & quality parameters
Throughput Studies
We help you select the perfect mix of prefilters and final filters by performing small scale filtration and determining the filtration area to reach your desired throughput
Extractables & Leachables
Our Extractables & Leachables testing study is a highly effective way to quantify and assess risks associated with leachable impurities from filters
Why Choose Us
With our extensive knowledge in membrane filtration and a wide range of products available at every stage of the process, along with our exceptional customer service, we are able to establish ourselves as the preferred partner.

Extensive Product Portfolio
At Nupore, we offer a wide range of filtration for your process requirements. Use our quick guide to land instantly at the product of your choice

Proven Membrane Technology

Fast Shipping
Talk To Us
Need assistance choosing the right product for your application, or have a question about ordering from us? Connect with an Expert